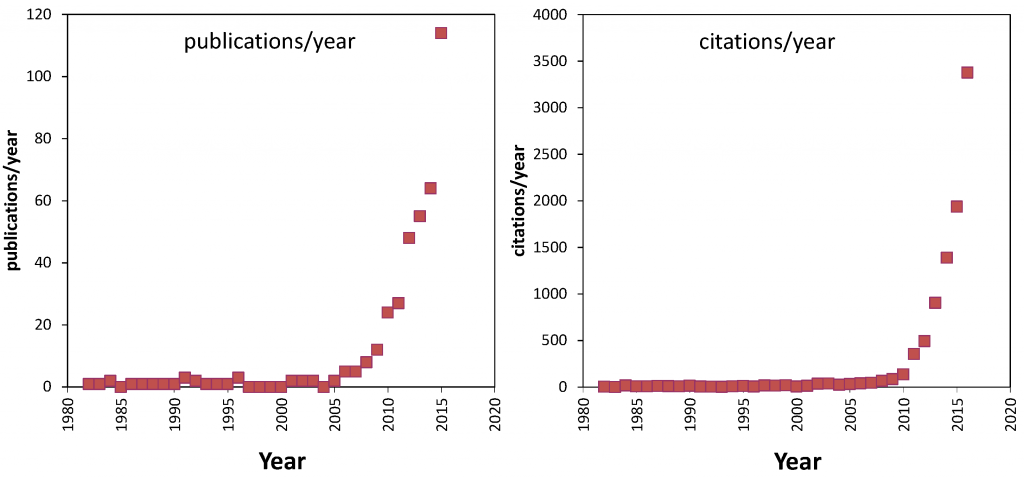
... and the field of Capacitive Deionization keeps on growing at an increasing speed ! With over 160 scientific papers written and published last year, the total number of CDI publications has grown from ∼25 in 2000, to ∼65 in 2010, to over 1,000 at the end of 2019! CDI papers in 2019 came from many prestigious places including Tsinghua University, Seoul National University, Technion, MIT, Stanford, and Yale. This output from such eminent research groups shows that CDI is taking a leading role in the scientific study of water desalination technologies. Analyzing these publication data for the past decade, an exponential growth can be observed, with a doubling of the publication output every 2.5 year !
Citations to the CDI literature have grown from a number less than 100 per year before 2010, to about 2000 per year at the end of 2015, and close to 10,000 per year in 2019. These statistics also reflect an exponential growth, with a doubling time of 2.0 year. This difference in doubling time (faster for citations than publications) may indicate that CDI papers are more and more cited in papers from outside the CDI-field.